Why the Potassium Sulfate Brine Process Is Gaining Popularity in Fertilizer Manufacturing

potassium sulfate (SOP) is one of the most used premium fertilizers. It is famous for its chloride free content with dual nutrient value. It is the preferred potassium fertilizer for chloride sensitive and premium produces like fruits, vegetables, tobacco, tea, and potatoes. The production of potassium is a complex process which needs robust machinery and energy. Historically, the manufacturing of SOP was dominated by the Mannheim process, a chemical reaction between potassium chloride and sulfuric acid. Compared with traditional production methods, the potassium sulfate brine process is attracting increasing attention from fertilizer manufacturers due to its lower production costs, reduced environmental impact, and higher product purity.
The potassium sulfate brine process is rapidly being adopted by the manufacturers because it combines high product quality, lower chloride contamination, and improved environmental controls. The advance technology from LANE Heavy Industry extract 90-95% potassium from the brine sources. The brine process utilizes mineral-rich brine resources or salt lake brines to extract potassium and sulfate compounds through evaporation, crystallization, separation, and granulation. The fertilizer manufacturers and investors who are seeking long-term profit while not harming the environment, adopting a modern potassium sulfate brine process production line from LANE Heavy Industry is a strategic choice.

What Is the Potassium Sulfate Brine Process?
The brine process is a manufacturing method that produces SOP fertilizer from potassium-rich natural brine, salt lake brine, seawater bittern, or industrial by-product brines.
Brine process is less energy intensive then Mannheim process, which uses potassium chloride and sulfuric acid at high temperatures to create potassium sulfate. The potassium sulfate brine process relies more on physical and chemical separation techniques such as:
- Fractional crystallization
- Double decomposition
- Flotation separation
- Drying and granulation
This process is suitable for regions with abundant salt lake resources, strong sunlight, and low rainfall conditions. The main output is high-purity potassium sulfate fertilizer containing potassium and sulfur, two essential plant nutrients.
Why demand is rising for the potassium sulfate brine process
- Chloride-sensitive crops and premium markets are expanding worldwide. This is increasing the demand for sulfate of potash (SOP); the brine process produces low-chloride SOP that meets these market needs.
- SOP supplies both potassium and sulfur in a single chloride-free product. This fertilizer improves crop quality and yield for fruits, vegetables, and other high-value crops. The brine process is economical and profitable which is driving adoption of production methods like the potassium sulfate brine process.
- Environmental and regulatory pressure is encouraging fertilizer makers to adopt processes that provide better gas and effluent control; brine-based routes can be designed to capture by-products and minimize emissions compared with older, less controlled methods.
Technical advantages of the potassium sulfate brine process
- Higher purity and consistent nutrient ratios: the brine process allows close control over solution composition and crystallization. Producing SOP with stable K2O and SO3 content meets premium grade specifications.
- Lower chloride and impurity carryover: by operating from concentrated brines and carefully controlled crystallization, the potassium sulfate brine process reduces chloride and insoluble residues in the final product. This is the key for chloride-sensitive markets.
The brine method can be integrated with modern reactors, crystallizers, and gas-treatment systems to create new by-product to increase revenue. This process is compatible with scrubbing and recovery systems to handle volatile acids and salts.
Understanding the Potassium Sulfate Brine Process
The technical core of the brine process involves several stages of mineral extraction and chemical transformation. It typically begins in solar evaporation ponds, where brine is concentrated to precipitate various salts in sequence.
- Solar Evaporation: Large-scale ponds use solar energy to evaporate water, concentrating the brine until minerals like halite (NaCl) and carnallite (KCl. MgCl2. 6H2O) crystallize.
- Fractional Crystallization: Through careful management of brine density, manufacturers isolate sulfate-bearing minerals such as schoenite (K2SO4. MgSO4. 6H2O) or leonite.
- Conversion and Flotation: These intermediate minerals are then reacted with potassium chloride (KCl) in a controlled aqueous environment. The potassium sulfate brine process uses the different solubilities of salts to precipitate K2SO4 crystals while keeping impurities in the “mother liquor.”
- Refining: The resulting crystals are washed and filtered to achieve a purity often exceeding 50% K2O content, with minimal chloride levels.
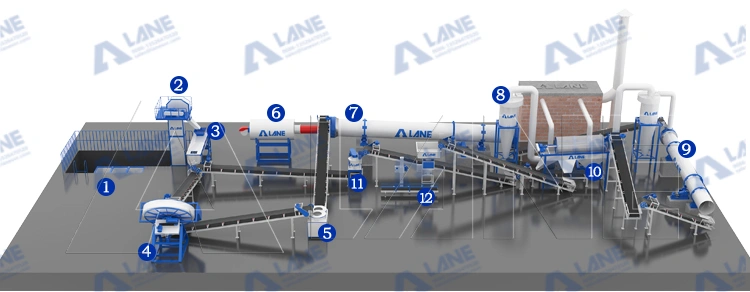
How LANE Heavy Industry’s fertilizer production line complements the brine route
LANE provide conversation tanks which is engineered for the chemical conversion and crystallization steps central to the brine process. We offer reactors, crystallizers, granulators, and downstream drying/cooling systems designed for stable SOP output.
Key equipment: feed preparation units, reactors, rotary drum granulators or disc granulators, rotary dryers, and automatic packing systems. These machines provide the material-handling and particle control needs of the brine process, enabling high throughput with uniform granule size.
LANE production line uses automation and emission control to provide precise control. Our gas and dust management provide safety from harmful substances. The result is a consistent premium product quality and regulatory compliance.
Process flow (brine-focused) practical overview
- Raw brine preparation: potassium-bearing brines, or dissolved potassium salts, are clarified and adjusted for composition before reaction or crystallization. The brine process starts here to ensure feed quality.
- Reaction/crystallization: controlled evaporation and cooling are used to precipitate potassium sulfate from the prepared brine; the brine process relies on optimized crystallizers to form desirable crystal habits and particle size distributions.
- Crushing: he raw SOP crystals from the brine process can often clump during the drying stage. LANE’s heavy-duty crushers ensure a consistent particle size before granulation.
- The Granulation System: LANE uses advanced Rotary Drum Granulators or Disc Granulators in this step to form round, durable granules. This is a critical step because granulated SOP prevents dust loss and ensures even nutrient distribution in the field.
- Drying and Cooling: After granulation the moisture content of the SOP produced must be controlled. LANE’s rotary dryers use high-efficiency heat exchange systems to dry the granules, followed by rotary coolers that stabilize the temperature to prevent caking during storage.
- Screening and Coating: To ensure the highest quality, LANE’s screening machines separate undersized and oversized particles for recycling. The final product is often treated in a coating machine to improve its flowability and moisture resistance.
- Automatic Packaging: Finally, the integrated production line includes automatic weighing and packaging systems, ensuring that the high-purity product from the potassium sulfate brine process is ready for the global market.
Economic and operational considerations
- Capital vs. operating expenditure: installing a potassium sulfate brine process integrated into a modern line like LANE’s requires higher initial investment than simple KCl-based lines, but producers often recover the cost through access to higher-priced SOP markets and improved process efficiency and reduced energy costs.
- Feedstock flexibility: the potassium sulfate brine process can be designed to accept various potassium sources (brines, kainite, KCl with conversion), giving plants flexibility when raw-material prices and availability change.
- Maintenance and corrosion management: acidic and saline streams require corrosion-resistant materials and careful maintenance; LANE emphasizes durable equipment and process design to reduce downtime for plants using the potassium sulfate brine process.
- Quality control: continuous monitoring of solution chemistry, crystallization parameters, and particle sizing is essential to keep SOP within specification—an area where the potassium sulfate brine process benefits from automated instrumentation on LANE lines.
Implementation tips for producers
- Conduct a raw-materials audit to determine whether brine feedstocks or conversion from KCl is most economic for your location; the potassium sulfate brine process can be adapted based on feedstock availability.
- Specify reactors, crystallizers, and material-contact components for corrosion resistance and ease of maintenance to maximize uptime for plants following the potassium sulfate brine process.
- Invest in automation and monitoring: real-time control of temperature, concentration, and residence time yields better SOP quality when using the potassium sulfate brine process.
- What is the potassium sulfate brine process?
The potassium sulfate brine process is a production method that extracts potassium sulfate fertilizer from potassium-rich brine using evaporation, crystallization, purification, drying, and granulation.
- Why is the potassium sulfate brine process better than the Mannheim process?
The potassium sulfate brine process generally offers lower energy consumption, lower emissions, and reduced raw material costs.
- What raw materials are used in the potassium sulfate brine process?
Common raw materials include:
- Salt lake brine
- Seawater bittern
- Potassium-rich industrial brines
- Can LANE provide a complete SOP production line?
Yes. LANE Heavy Industry provides complete equipment and turnkey solutions for the potassium sulfate brine process.

Of course, we also offer other types of products for your different needs, you can click to view.
For more information, please contact us by sending an inquiry today! We can help you!






Write a Reply or Comment