SOP Fertilizer Acid Reaction Method: Equipment, Conditions, and Yield Improvement

Sulfate of Potash (SOP, K₂SO₄) is a premium potassium fertilizer valued for its low chloride and dual nutrient value. This fertilizer provides both potassium and sulfur in high quality and quantity, which makes it indispensable for highvalue, chloridesensitive crops such as tobacco, citrus, potatoes and greenhouse vegetables. The SOP fertilizer acid reaction method has become one of the most reliable industrial processes for producing high-purity sulfate of potash. The SOP fertilizer acid reaction method uses the Mannheim process, and it dominates global production, accounting for 50–60% of supply.
This article explores the SOP fertilizer acid reaction method in depth, examining the core equipment, critical operating conditions and proven strategies for yield improvement within an SOP fertilizer acid reaction line provided by LANE Heavy Industry Machinery Technology Co., Ltd.
Process of the SOP Fertilizer Acid Reaction Method Chemistry
The SOP fertilizer acid reaction method involves reacting potassium chloride (KCl) with sulfuric acid (H₂SO₄) in a hightemperature furnace to produce potassium sulfate (K₂SO₄) and hydrogen chloride (HCl) gas. The simplified reaction can be expressed as:
KCl + H₂SO₄ → K₂SO₄ + HCl
In practice, the reaction occurs in two steps: first formation of potassium bisulfate (KHSO₄), followed by its conversion to K₂SO₄ with additional KCl. The hydrogen chloride gas can be turned into hydrochloric acid (HCl) for industrial use. This process is widely used due to its ability to produce high-purity SOP with controlled particle size and minimal impurities. However, achieving optimal results requires precise control over reaction conditions and equipment.
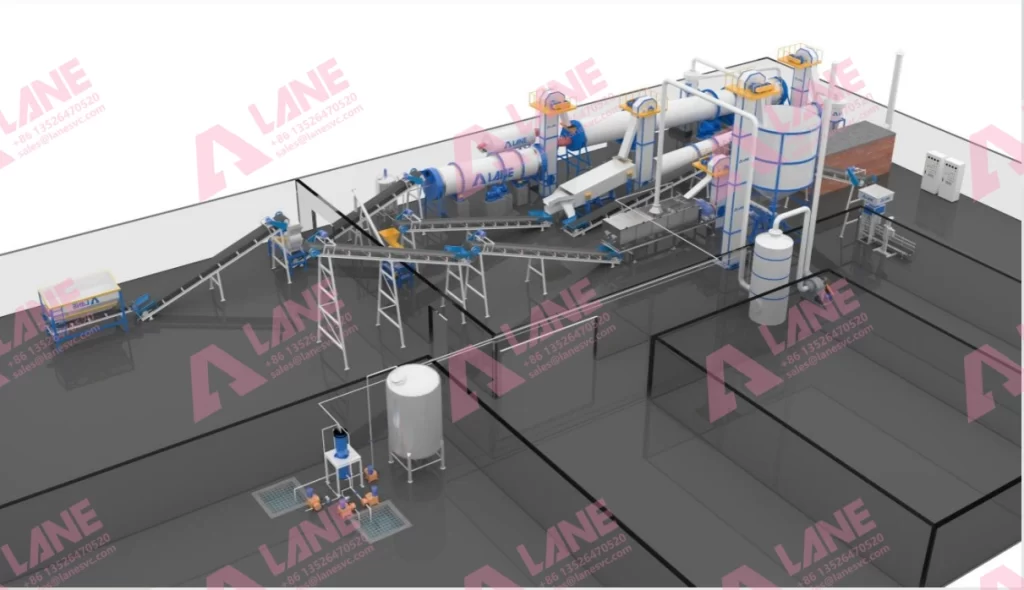
Core Equipment in the LANE Heavy Industry Production Line
Modern SOP production lines designed and engineered by LANE Heavy Industry integrate multiple systems and ensure each unit is engineered for compatibility and maximum efficiency. Key equipment includes:
Acid Reaction Furnace: The acid reaction furnace is the heart of the SOP fertilizer acid reaction method. It is a specialized, refractorylined furnace in which KCl and H₂SO₄ are intimately mixed at 500–600 °C. LANE’s furnace is constructed from thermal and corrosionresistant materials capable of withstanding up to 2000 °C, ensuring long service life and high reaction yield.
Sulfuric Acid Storage & Dosing System: Highconcentration acid (>80%) is stored in dedicated tanks with safety interlocks and precisely metered into the furnace via automated control valves. This automatic dosing system ensures the correct ratio of KCl to sulfuric acid; this removes human error and improves consistency.
Gas Absorption System: The reaction produces hydrogen chloride gas. LANE’s system safely captures and treats the hydrogen chloride gas, turning it into hydrochloric acid. We turn a byproduct into a valuable secondary product while minimizing environmental impact.
Cooling Systems: Hot SOP crude product exits the furnace into an enclosed cooling drum.
Crushing & Grinding Units: A primary crusher reduces the cooled cake to manageable chunks; a chain mill or hammer mill then grinds it to the desired powder fineness (typically 90% passing 60 mesh).
Dust Collection: Cyclone dust collectors capture airborne particles and reintroduce them into the production stream.
Granulation (optional): For granular SOP, LANE adds double roller or rotary drum granulators that form uniformly sized, easytohandle granules.
Automatic Packaging & PLC Control: The entire SOP fertilizer acid reaction method is controlled by a central PLC system that monitors temperature, feed rates and equipment status in real time.
Critical Process Conditions
Yield and product quality are exquisitely sensitive to the following parameters:
| Parameter | Optimal Range | Influence |
| Reaction temperature | 500–600 °C | Ensures complete conversion; too low gives unreacted KCl, too high increases byproducts |
| Acid concentration | 93–98 % H₂SO₄ | Minimizes water load, reducing energy cost and corrosion |
| Residence time | 20–40 min (depending on furnace design) | Allows the twostep reaction to reach equilibrium |
| KCl particle size | <2 mm | Promotes rapid, uniform reaction |
| Gas extraction | Continuous HCl removal | Drives equilibrium toward K₂SO₄ and recovers valuable HCl |
LANE’s proprietary control system maintains these variables within narrow limits, ensuring a consistent product with K₂O content of 50–52 %, sulfur >17 % and chloride <1.5 %.
Strategies for Yield Improvement
Achieving high yield and purity in the SOP fertilizer acid reaction method depends heavily on maintaining optimal process conditions. It involves both reactorlevel and plantwide measures:
- Temperature Profiling: The reaction typically occurs at elevated temperatures (above 500°C). Maintaining stable heat ensures complete conversion and prevents side reactions. Our advanced furnace designs employ multizone heating to optimize the temperature ramp and boost conversion rates.
- Precision Feeding: The balance between potassium chloride and sulfuric acid must be carefully controlled. Excess acid can lead to corrosion and waste, while insufficient acid reduces conversion efficiency. Gravimetric lossinweight feeders guarantee the exact stoichiometric ratio of KCl to H₂SO₄, eliminating waste.
- Enhanced Mixing: Advanced furnaces from LANE Heavy Industry are designed to optimize residence time without sacrificing throughput. Internal baffles and agitators in the furnace improve contact between solid and liquid reactants. This shortens the required residence time. Adequate reaction time is necessary for full conversion.
- Corrosion Resistant Materials: The inner lining of the furnace is built using refractory materials that can withstand temperatures above 700°C. Typical refractory materials include:
- Highalumina bricks
- Silica bricks
- Fire clay bricks
- Castable refractory cement
ClosedLoop Recycling: All dust and offspec material is returned to the process, raising raw material utilization to >99 %.
Advantages of Using LANE Heavy Industry Machinery
LANE Heavy Industry’s fertilizer production lines are specifically designed to support the SOP fertilizer acid reaction method with high efficiency and reliability. Key advantages include:
- Durable construction materials resistant to acid corrosion
- Integrated systems that streamline production from raw material input to final packaging
- Energyefficient designs that reduce operational costs
- Customizable configurations to match different production capacities
- Strong technical support and aftersales service
By integrating these advanced systems, manufacturers can achieve higher productivity while maintaining strict environmental and quality standards.

Environmental and Safety Considerations
The SOP fertilizer acid reaction method involves handling high temperatures and corrosive materials, making safety and environmental protection essential. Modern systems incorporate:
- Sealed reactors to prevent gas leakage
- Scrubbing systems for HCl gas
- Automated monitoring and sensors to detect abnormalities
- Waste heat recovery systems to reduce energy consumption
LANE Heavy Industry emphasizes ecofriendly designs that align with global environmental regulations, helping producers minimize their carbon footprint.
FAQ (SOP Fertilizer Acid Reaction Method)
Q1: What is the typical yield of the SOP fertilizer acid reaction method?
A: With LANE’s optimized line, potassium recovery reaches 95–97%, and SOP product purity is ≥98% (0-0-52 grade).
Q2: How does LANE’s reactor solve corrosion issues in the acid reaction method?
A: LANE uses Inconel 625 hightemperature alloy for the reactor lining, which resists sulfuric acid and HCl corrosion at 700°C, extending service life to 15+ years.
Q3: What byproduct is generated, and is it usable?
A: The SOP fertilizer acid reaction method produces HCl gas, which LANE’s system recovers as 30–33% industrial hydrochloric acid, a saleable byproduct that improves project economics.
Q4: Can the line handle variable raw material quality?
A: Yes. LANE’s PLC dosing system autoadjusts the KCl:H₂SO₄ ratio based on realtime feed analysis, maintaining stable yield even with minor raw material variations.
Of course, we also offer other types of products for your different needs, you can click to view.
For more information, please contact us by sending an inquiry today! We can help you!






Write a Reply or Comment